Products
Evolution-RO™ System
-
Our best-selling High-Flow system just got better! Improved Dry Membrane design now with 20% increase in flow rate. Up to 1,200+ GPD!
- Capable of producing up to 1,200 GPD (gallons per day) or 50+ GPH (gallons per hour) of pure water, perfect for medium to large hydroponic operations (Flow rates of RO systems are based on source water @ 65 psi/77°/500 ppm)
- Lowers parts per million (ppm) of total dissolved solids (TDS) by up to 95%
- SAVE MORE WATER by running a 1:1 waste to product water ratio
- Includes ChloraShield™ carbon filter for up to 100% chloramine removal
- Dry High-Flow Reverse Osmosis membranes (x2)
- Inlet pressure gauge with safe zones—liquid filled
- Garden hose connector
- 1:1 and 2:1 ratio restrictors
- Comes with:
- 6' length, white inlet tubing
- 10' length, blue purified water outlet tubing
- 10' length, black wastewater drain tubing
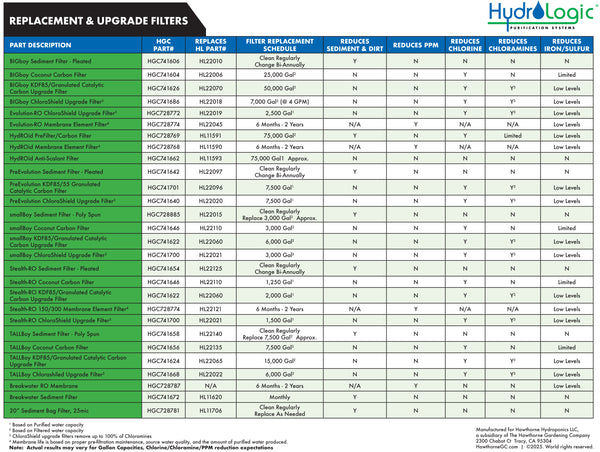
- Evolution-RO filter housings must be replaced every 3 years
- Replaces HL 31023
- Dimensions (H x W x D): 13" x 23" x 13"
- Weight: 21.40 lbs
- Capable of producing up to 1,200 GPD (gallons per day) or 50+ GPH (gallons per hour) of pure water, perfect for medium to large hydroponic operations (Flow rates of RO systems are based on source water @ 65 psi/77°/500 ppm)
-
Filters & Replacement Schedules
ChloraShield® Filter- Replace after 2,500 gal (Filter replacement schedules for RO carbon filters based on product water)
- Replace between 6 months–2 years
-
Evolution-RO™ System Downloads
Part Number: HGC728992

Q: How do I properly store a Reverse Osmosis (RO) System that won't be used for more than 3 weeks
A: Reverse osmosis membranes like water to run through them often in order to perform properly. Running water for once a day for a short period of time is ideal, but even once a week is sufficient. If you are not going to use your RO system for 3–4 weeks or longer, there are storage procedures you should follow to maintain optimum system performance. Take the membrane(s) out of the system and place in a plastic bag and put them in the refrigerator. A white plastic kitchen bag with a knot tied at the top works well. Take the carbon filter (and in some systems such as the Stealth-150/300 the sediment filter as well) out of the housing and let it (them) dry out. Leave the housings and the caps on the system unscrewed to let them dry out. The purpose of this is to not trap wet filters and moisture in the system so that bacteria will not grow. As an extra measure of precaution, sterilizing using a light bleach or hydrogen peroxide solution to clean the housings, tubing and fittings is a good idea before using the system again.
Q: Can the Evolution-RO be used as a drinking water system for my home?
A: The Evolution is not really designed to be an under the sink drinking water system. The reason for this is because the large membranes in that system perform what is called “TDS (total dissolved solids) creep.” This means upon system start up the PPMs (parts per million) will come out higher for a minute or two and then go back down to the standard rejection %. This happens when there is untreated water in the membranes/housings from the last time you ran the system and then shut it off. When filling a 50 or 100 gallon reservoir this really is insignificant as it evens out with the rest of the purified water. One option for using the Evolution for drinking water is to use it to fill 5 gallon jugs and put them on a water dispenser (like the ceramic ones).
Q: How does RO affect pH?
A: RO product water can have a lower pH than the feed water. It depends on the level of dissolved CO2 in the feed water. A lowering of pH can occur because the existing CO2 in the water, being a gas, passes through the membrane but the alkaline constituents, primarily HCO3 (bicarbonate), are removed as part of the reject water. In the feed water the HCO3 neutralizes the acid-causing CO2. In the product water, with HCO3, greatly reduced, the CO2 is free to combine with the H2O to form carbonic acid: CO2 + H2O > H2CO.
Another reason to question the pH reading is that for any water sample which is very low in TDS it is very hard to obtain an accurate pH value. The worst case of this accuracy problem occurs with de-ionized water but some low TDS RO water will react the same. The sample is so low in minerals that there is no buffer. The slightest absorption or loss of CO2 shifts the pH substantially. The water’s tendency to pick up additional CO2 from the ambient air, even if the test is done on site, can produce a very low false pH.
In reality, measuring the pH of RO water is difficult and not accurate due to lack of any pH buffering the RO water. If the feed water has a lot of dissolved CO2 then pH can drop a few points. But, in the case of a drinking water RO system, a post re-mineralization filter will help neutralize the final product water’s pH level. In the case of RO water for horticultural purposes, once you mix a calcium/magnesium supplement into the RO water and then add your nutrients and additives, the final pH can be adjusted and your feed formula should be pH stable.

Q: I have well water, what challenges can I expect?
A: Here are the most common challenges when filtering well water:
- It is typically colder which will decrease your flow rates because cold water flows slower through a reverse osmosis membrane
- Often has higher PPM/TDS levels (hardness) which can decrease the life of a membrane
- Can contain high levels of sediment which can easily clog a carbon or sediment pre-filter
- Can have high levels of iron, manganese and hydrogen sulfide (rotten egg smell)
- It occasionally contains iron bacteria which is difficult to remove
- It generally has low water pressure
Q: How do I treat water containing iron?
A: There are different methods of treating iron depending on how much iron is present in the source water. The easiest way to know how much iron you have is to get your water tested from NTL or a reputable testing laboratory. Once you have your water analysis, please contact us with your water report and we can assist you with choosing an appropriate prefiltration method.
Q: How do I know if I have iron in my water?
A: If your carbon or sediment filter appears to be red, maroon, orange or brown in color you likely have iron in your water. You may also notice that your sinks and or toilets also have this discoloration. See an example image below of a system that was used to filter water with excessive iron levels.

Q: Why do pH levels come out higher than the source water when running water through a carbon or Carbon/KDF filter?
A: This is common, there is generally a significant pH spike from the GAC (granular activated carbon) that is mitigated relatively quickly after start up. For some systems, it can take days or even weeks to run enough rinse water to get the pH back to normal. Since it is temporary, it is generally not a significant issue. For more information contact HydroLogic.












 Dechlorinators by HydroLogic
Dechlorinators by HydroLogic How to Operate Quick Connect (QC) Fittings
How to Operate Quick Connect (QC) Fittings How to Calculate the Flow Rate of Your Reverse Osmosis System
How to Calculate the Flow Rate of Your Reverse Osmosis System Evolution-RO: How to Install The Continuous Duty Booster Pump
Evolution-RO: How to Install The Continuous Duty Booster Pump